All published articles of this journal are available on ScienceDirect.
Oxidative Stress and Antioxidant Imbalance in Erythema Multiforme: A Cross-sectional Study of Salivary Biomarkers
Abstract
Introduction
This cross-sectional study aims to assess levels of antioxidants, including Superoxide Dismutase, Glutathione Peroxidase, Catalase, and Uric acid, in the saliva of patients with erythema multiforme. The saliva samples were collected during the active stage of the disease, approximately 2–4 days after lesion onset. Additionally, correlations were examined to assess differences in antioxidant levels between patients with recurrent episodes and those with a single episode of the disease. An independent t-test was utilized to compare controls with patients, and Pearson correlation analysis was performed for subgroup comparisons.
Methods
A total of 45 patients and 45 controls participated in the study. Saliva samples were collected to determine levels of the above antioxidants in both groups. The presence of typical target skin, in addition to a previous prodrome of moderate fever, is considered a diagnostic feature. The patient group was divided into two subgroups: the first subgroup included patients with recurrent EM episodes in the previous year, and the second subgroup included patients with a single episode.
Results
The calculated levels of all measured salivary antioxidants were lower in patients with erythema multiforme than in the control group, with a significant difference in catalase (P-value < 0.05) and a highly significant difference in glutathione peroxidase, uric acid, and superoxide dismutase (P-values < 0.001). The group of patients was further subdivided into those with a single episode of erythema multiforme and those with multiple episodes; the difference was insignificant for all measures except catalase, which was significantly lower in the multiple-episode subgroup (P-value < 0.05).
Discussion
This study revealed an important difference in antioxidant levels between the control group and EM patients. Definitely, the salivary concentrations of SOD, GSH-Px, and UA in EM patients were particularly lower compared to those in the control group, suggesting that EM patients have a compromised antioxidant defense.
Conclusion
The salivary secretion of erythema multiforme patients demonstrated a modified antioxidant status, characterized by lower levels of these markers compared to those of healthy individuals. These findings reflect association rather than causation due to the cross-sectional design, and longitudinal validation is required. The difference in Catalase levels between the single-episode and multiple-episode erythema multiforme subgroups may reflect differences in oxidative-stress dynamics in recurrent disease, warranting further investigation.
1. INTRODUCTION
Erythema multiforme (EM) is an immune-mediated, acute medical case that primarily affects the skin and mucous membranes, with oral mucosal involvement in many cases, and can occur after a herpes simplex virus infection or as a reaction to certain drugs. The exact pathogenesis of EM is not fully recognized; however, it may be related to an immunological response that triggers the release of inflammatory mediators, resulting in oxidative stress and tissue damage, manifested as a sudden onset of erythematous target lesions on the skin and ulcerative and erythematous lesions in the oral cavity with distinguishable lip crustation [1]. In this study, EM cases were diagnosed clinically and included regardless of the underlying etiology (HSV-associated, drug-related, or idiopathic), as the focus was on disease expression rather than causal classification.
Oxidative stress has been considered a contributing factor in numerous oral lesions, including recurrent aphthous stomatitis, oral lichen planus, and Behcet’s disease [2-4]. Factors such as immune dysregulation, stress, and infection can disrupt the balance between oxidants and antioxidants (AOs), leading to increased free radical accumulation and contributing to cellular damage and inflammation [5].
AO mechanisms, including enzymes such as catalase (CAT), glutathione peroxidase (GSH-Px), and superoxide dismutase (SOD), are significant in neutralizing these radicals and preventing further tissue injury [6]. Uric acid (UA) also serves as a potent antioxidant, scavenging reactive oxygen species and contributing to reduced oxidative stress [7].
Recent studies suggest that human saliva has a unique AO system, including uric acid, CAT, GSH-Px, and SOD, with uric acid contributing significantly to the overall AO capacity of saliva [8]. To the best of the authors’ knowledge, no previous studies up to 2024 have specifically evaluated oxidative-stress biomarkers in the salivary secretion of EM patients, despite increased interest in oxidative markers in other inflammatory mucosal diseases.
Given this knowledge gap, this study aims to investigate whether an antioxidant imbalance in saliva may contribute to EM pathophysiology. We hypothesized that salivary antioxidant enzyme levels are lower in EM patients than in healthy individuals. Additionally, we expected a greater reduction in antioxidant levels among patients with recurrent EM, suggesting a possible link to disease recurrence.
2. PATIENTS AND METHODS
2.1. Participants and Study Design
Forty-five patients diagnosed with erythema multiforme and 45 healthy controls were included in this study from February 2024 to June 2024, as agreed by the scientific research and ethical committee at the Oral Pathology Department, Kufa University’s College of Dentistry, with ethics number 3F24, and followed the Helsinki Declaration. The estimated sample size was determined using Master’s software (CMC, Vellore) to be 45 for each group. This determination was based on a mean difference of 1.5 with an effect size of 0.8, while maintaining a 5% alpha error and 80% power. Potential technical errors were considered, with an anticipated dropout rate of approximately 10–15%.
Participants were recruited using convenience sampling.
Whole unstimulated saliva was collected during the active disease phase, approximately 2–4 days from lesion onset.
2.2. Inclusion Criteria
The controls were matched to the patient group for age and sex. All participants were asked to sign a consent form before salivary sample collection. The inclusion of patient group members was based on clinical features, including generalized ulcerative and erythematous lesions of the oral mucosa and lip crustation, all of which were diagnosed as EM. The presence of typical target skin, in addition to a previous prodrome of moderate fever, is considered a diagnostic feature. The patient group was divided into two subgroups: the first group consisted of patients with recurrent EM episodes in the last year, and the second group consisted of patients with a single episode. Etiology was not used as a classification criterion; EM cases were included regardless of underlying cause (HSV-associated, drug-induced, idiopathic).
2.3. Exclusion Criteria
The factors that were excluded in this study include: smokers, alcoholics, pregnant women, and patients with a history of other inflammatory and immune-related diseases, or those receiving prolonged systemic medications such as corticosteroids, antivirals, antibiotics, or immunosuppressants during the last two months. Before saliva collection, all participants were asked to rinse their mouths with water. After that, each one was asked to provide 5 mL of total unstimulated human saliva in a plastic tube. Samples were placed directly on ice and stored at −20°C within 30 minutes. Only one freeze–thaw cycle was permitted before analysis.
2.4. Biochemical Procedures and Analysis
The Marklund method was employed to evaluate SOD levels [9]. For GSH-Px analysis, the procedure suggested by Paglia DE and Valentine WN was used [10]. The CAT action was evaluated by the approach outlined by Cimen et al. [11]. The UA activity was measured using the procedure by Fossati et al. [12, 13].
Assays were performed using commercial reagent sets (manufacturer: Spectrum Diagnostics, Cairo, Egypt; SOD Kit Cat. No. 252-001; GSH-Px Cat. No. 253-002; CAT Cat. No. 254-003; UA Cat. No. 248-001). All assays were performed once per sample (no duplicates), and the manufacturer did not provide inter- or intra-assay coefficients of variation. Laboratory personnel were blinded to the participant group (patient/control) to minimize analytical bias. Salivary antioxidant levels were normalized to total protein concentration measured using the Bradford assay (Bio-Rad, USA), and mean salivary total-protein values were comparable between groups.
2.5. Analysis of Data
The statistical analysis to evaluate differences in antioxidant levels in saliva between the EM and control groups employed an independent t-test. Additionally, the relationship between AO levels in the two subgroups was analyzed using Pearson correlation. Data were tested for normality using the Shapiro–Wilk test before applying parametric analyses. Due to the exploratory nature and limited number of biomarkers, no adjustment for multiple comparisons was applied; however, p-values are reported exactly to allow interpretation. Salivary flow rate was within the normal expected physiological range in participants; therefore, no adjustment for flow rate was applied, but this is acknowledged as a potential influence in interpretation.
3. RESULTS
The study included two groups: patients and a control group, each comprising 45 participants (21 males and 24 females). For age distribution, the mean age of the patients was 30 years (18-43), while the control group had a mean age of 34 years (18-48) (Table 1).
| - | Age (Years) | Sex | ||
|---|---|---|---|---|
| Means | Range | Male | Female | |
| EM (n=45) | 30 | 18-43 | 21 | 24 |
| Controls (n=45) | 34 | 18-48 | 21 | 24 |
The SOD grade in the patient group's saliva was lower than in the control group (mean difference = 0.59, 95% CI: 0.43–0.75, effect size = 1.51, P < 0.001). Similarly, both GSH-Px and UA were significantly reduced in patients compared to controls (GSH-Px mean difference = 0.62, 95% CI: 0.51–0.73, effect size = 2.63, P < 0.001; UA mean difference = 1.62, 95% CI: 1.42–1.82, effect size = 3.48, P < 0.001). For CAT, the difference was smaller but still significant (mean of difference = 0.29, 95% CI: 0.15–0.43, effect size = 0.87, P = 0.02), with controls showing higher levels (Fig. 1 and Table 2).
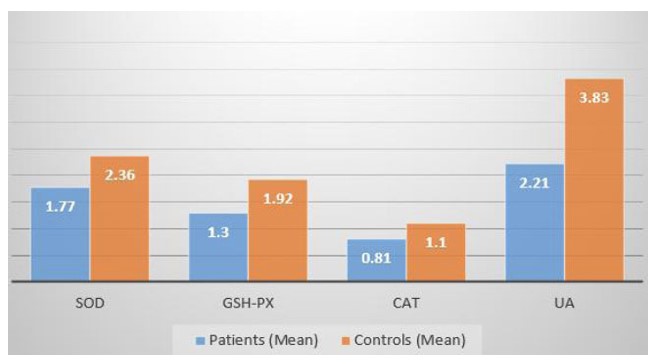
Comparison between the levels of AOs between the control and the patient groups. SOD = Superoxide Dismutase. GSH-Px = Glutathione Peroxidase. CAT = Catalase. UA =Uric acid.
| - | EM (n=45) | Controls (n=45) |
CL (Effect Size) |
P-value |
|---|---|---|---|---|
| SOD (U/mg Protein) (Mean±SD) | 1.77±0.23 | 2.36±0.49 | 0.43-0.75 (1.51) |
<0.001* |
| GSH-Px (U/mg Protein) (Mean±SD) |
1.29±0.22 | 1.92±0.28 | 0.51-0.73 (2.63) |
<0.001* |
| CAT (U/mg Protein) (Mean±SD) |
0.81±0.23 | 1.103±0.40 | 0.15-0.43 (0.87) |
0.02* |
| UA (mg/dl) (Mean±SD) |
2.209±0.264 | 3.83±0.602 | 1.42-1.82 (3.48) |
<0.001* |
The salivary concentration of SOD in the single episode EM subgroup was approximately equal to that in the multiple episodes EM subgroup. For GSH-Px and UA, the salivary levels were slightly higher in the single-episode group of EM than in the numerous episodes group of EM. However, there was no significant difference (P-value >0.05), while the CAT salivary level of the single-episode EM was significantly higher (P-value <0.05) than that of the second group (Table 3). A post-hoc power calculation based on the observed effect size for catalase indicated approximately 70–75% statistical power to detect differences between subgroups, suggesting limited robustness for this outcome.
| - | SOD (U/mg Protein) | GSH-Px (U/mg Protein) | CAT (U/mg Protein) | UA (mg/dl) |
|---|---|---|---|---|
| Single episode (n=29) |
1.757± 0.222 |
1.32± 0.146 |
0.885± 0.128 |
2.259± 0.207 |
| Multiple episodes (n=16) | 1.79± 0.272 |
1.249± 0.32 |
0.676± 0.323 |
2.118± 0.334 |
| P-value | 0.653 | 0.372 | 0.023* | 0.139 |
4. DISCUSSION
This study introduces the first evidence, to our knowledge, of a mediated salivary AO system for patients with Erythema Multiforme (EM). The results indicated decreased levels of non-enzymatic (UA) AOs and salivary enzymatic (SOD, GSH-PX, CAT) in those EM patients compared to healthy controls. Additionally, this study found that patients with recurrent EM episodes have lower catalase levels than those with a single attack. The global result suggests that the disturbances in the environments of AOs could be an essential setting of EM`s pathophysiology, affecting even the disease recurrence.
The effect size scales indicate that the differences in AO levels between controls and patients are not only statistically significant but also clinically meaningful, particularly for UA (effect size 3.48) and GSH-Px (effect size 2.63), which indicate large effects. The narrower confidence intervals across all markers strengthen the reliability of these results, confirming that the reduced AO levels in EM patients are consistent and unlikely to be due to random variation.
These findings suggest a possible clinical association between salivary AOs and EM. The constant decrease in SOD, UA, and GSH-Px levels suggests that these biomarkers could help as non-invasive tools for early diagnosis, monitoring disease recurrence, and potentially predicting EM severity.
The importance of the SOD enzyme lies in its role in changing free radicals into less harmful products, such as hydrogen peroxide [14, 15]. The reduction in this enzyme compromises defense against oxidative stress, leading to cellular damage and tissue inflammation. Furthermore, reduced levels of UA and GSH-Px in the salivary secretion of EM patients may result in a lower capacity to neutralize reactive oxygen species (ROS), thereby leading to greater oxidative damage [16]. The high reduction in these antioxidants refers to a general decline in the primary defence mechanism against ROS. The first line of defence is SOD, which catalyses the dismutation of superoxide radicals into hydrogen peroxide; thereafter, the other AOs participate in the subsequent degradation of H2O2 into water and oxygen [17].
A study conducted by Shirazi and colleagues measured salivary AO levels in patients with oral lichen planus. They found no statistical difference between UA levels between patients and the control group; however, for GSH-Px and SOD levels, the elevated salivary concentrations in healthy participants were significantly different [18]. This may reflect a major difference in the chronic T-cell-mediated nature of OLP compared to the acute immune-complex- or cytokine-related hypersensitivity in EM.
Our findings align with studies on recurrent aphthous ulceration, where Jesija et al. also documented salivary AO and plasma levels in patients with recurrent aphthae, found that disturbances in these enzymes were present in those patients, and that this disturbance was correlated with the severity of the ulceration [19]. This similarity reveals a common antioxidant pathway in the pathophysiology of acute oral ulcerations.
This study also examined salivary AOs levels in both single and multiple EM episodes. The findings suggest that the frequency of EM episodes may not directly reflect the total levels of these AOs. However, CAT degrees were significantly higher in the group with a single episode, suggesting that patients with their first experience of the disease have a more robust AO, probably due to an acute immune reaction [20].
While the decreased levels of CAT in those experiencing multiple attacks could be attributed to the overwhelmed AO defense, which will lead to a minimized response caused by repeated occurrences. This may result from prolonged immune activation or from the persistent nature of tissue inflammation, leading to adaptive changes in AO function [21]. Moreover, the chronic high level of H2O2 in the recurrent episodes condition may lead to the inactivation of the catalase enzyme through a cyclic redox reaction, resulting in a vicious cycle when the defence against H2O2 is weak, lowering the threshold in subsequent episodes.
Many previous studies have highlighted a positive correlation between EM and prior herpes simplex viral infection, which is considered the main predisposing factor [22, 23]. The decline in AO activity in those patients could be attributed to the direct effect of the virus, which increases the oxidative stress and lipid peroxidation [24].
Due to the cross-sectional design, causality between oxidative imbalance and EM cannot be established, and the results reflect only a single time point. Uncontrolled factors such as oral hygiene and recent systemic conditions may have influenced salivary biomarkers. The small recurrent-episode subgroup (n = 16) provided moderate statistical power (~70–75% for catalase), which limited subgroup interpretation. Minor pre-analytic and assay variability should also be considered. Therefore, findings should be interpreted cautiously and validated in longitudinal studies.
5. LIMITATIONS
The study limitations include the short observational period, which is cross-sectional and insufficient to establish a causal relationship between the onset of EM and AO levels; therefore, longitudinal studies are essential to determine over-time changes in AO levels and their contribution to the pathophysiology of this disease. Furthermore, saliva collection occurred within a narrow time window (2–4 days from lesion onset), during the active disease phase, which may introduce timing-related variability. Possible interference factors-such as recent systemic infections and individual oral hygiene status-were not recorded and may have influenced salivary biomarker concentrations. The recurrent-episode subgroup had a relatively small sample size (n = 16), providing only moderate post hoc statistical power (~70–75% for catalase), which may border on the interpretability of subgroup analyses. Additionally, the use of a single freeze–thaw cycle and the absence of detailed assay reproducibility metrics (intra- and inter-assay CVs) represent further methodological restrictions.
Furthermore, additional studies may include more indicators of oxidative stress in the AO estimation, such as F2-isoprostanes or malondialdehyde (MDA), to provide a deeper understanding of the oxidative landscape in EM.
CONCLUSION
The salivary secretions of patients with EM exhibit different AO status, with lower levels of CAT, GSH-Px, UA, and SOD than those of healthy persons. Because of the cross-sectional design of this study, these results indicate an association rather than a causal relationship between oxidative stress imbalance and EM. The difference in CAT levels between the single- and multiple-episode EM subgroups further suggests a potential role of AO defenses in disease recurrence. However, these findings should be considered preliminary, and longitudinal studies are required to explain the temporal relationship between oxidative alterations and disease progression. Overall, this study's results highlight the importance of monitoring oxidative stress markers in EM patients, which may support early detection, improve disease management, and potentially reduce recurrence.
AUTHORS’ CONTRIBUTIONS
The authors confirm contribution to the paper as follows: K.M., A.A. and A.M.: Conceptualization, Review-writing & editing; K.M. and A.M.: Data collection; A.A.: Data analysis. The authors have read and approved the published form of this manuscript.
LIST OF ABBREVIATIONS
| EM | = Erythema Multiforme |
| SOD | = Superoxide Dismutase |
| GSH-Px | = Glutathione Peroxidase |
| CAT | = Catalase |
| UA | = Uric Acid |
| AO | = Antioxidant |
| ROS | = Reactive Oxygen Species |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
This cross-sectional study was approved by the scientific research and ethics committee of the Oral Pathology Department at Kufa University's College of Dentistry, Iraq with ethics number 3F24.
HUMAN AND ANIMAL RIGHTS
All procedures performed in studies involving human participants were in accordance with the ethical standards of institutional and/or research committee and with the 1975 Declaration of Helsinki, as revised in 2013.
CONSENT FOR PUBLICATION
Informed consent was obtained from all patients who participated in the study.
AVAILABILITY OF DATA AND MATERIALS
The data supporting the findings of the article is available in the Zenodo Repository at (https://zenodo.org/uploads/18646676).
ACKNOWLEDGEMENTS
Declared none.


