All published articles of this journal are available on ScienceDirect.
Comparative Efficacy of Simvastatin-impregnated Gel Foam and Platelet-rich Fibrin (PRF) for Alveolar Ridge Preservation Following Mandibular Molar Extraction: A Prospective Randomized Controlled Clinical Trial
Abstract
Introduction
This study aimed to compare the regenerative potential and soft tissue healing outcomes of Simvastatin-impregnated gel foam and Platelet-Rich Fibrin (PRF) following mandibular molar extraction.
Methods
Forty patients undergoing mandibular molar extraction were randomly allocated into two groups: Simvastatin-impregnated gel foam (Group A) and PRF (Group B). Outcomes assessed included postoperative pain (Visual Analog Scale, VAS), soft tissue healing (Landry’s scale), bone regeneration (Cone Beam Computed Tomography, CBCT), and postoperative complications. Statistical analyses included t-tests and Chi-square tests.
Results
Demographic characteristics were comparable between groups. The mean extraction times were similar (p =0.67). Postoperative pain scores decreased over time without significant differences between groups (p >0.05). Soft tissue healing scores showed no significant differences across time points (p >0.05). However, bone density at three months postoperatively was significantly greater in the Simvastatin group (422±30 HU) than in the PRF group (411±28 HU) (p =0.01). Minimal postoperative complications occurred, with slightly fewer in the Simvastatin group.
Discussion
Simvastatin-impregnated gel foam showed superior bone regeneration compared to PRF in alveolar ridge preservation after mandibular molar extraction. Both materials effectively reduced postoperative pain and improved soft tissue healing. Simvastatin’s enhanced osteogenic, anti-inflammatory, and antimicrobial effects indicate it as a promising, minimally invasive option for superior bone healing and socket preservation.
Conclusion
Both Simvastatin-impregnated gel foam and PRF effectively promoted alveolar ridge preservation, though Simvastatin demonstrated superior bone regenerative capabilities.
1. INTRODUCTION
Alveolar bone significantly influences dental integrity, periodontal health, and facial esthetics, serving as an essential structural component that supports teeth via the periodontal ligament and effectively dissipates masticatory forces. Tooth extraction triggers a cascade of physiological events, initiating alveolar bone remodeling characterized by resorption, predominantly in horizontal and vertical dimensions. This resorption can result in a loss of approximately 40–60% of alveolar ridge width within 6 to 12 months post-extraction, significantly impacting subsequent prosthetic rehabilitation and implant placement outcomes, as well as altering facial esthetics and functional occlusal relationships [1-3].
Traditional alveolar ridge preservation strategies have predominantly relied upon bone grafting techniques employing various graft materials, including autologous, allogeneic, xenogeneic, and synthetic grafts, often in conjunction with barrier membranes or growth factor-based therapies. Despite their widespread use, these approaches frequently have inherent limitations, including unpredictable graft resorption rates, potential risk of disease transmission, donor site morbidity, increased procedural complexity, and handling difficulties [4-6]. Consequently, there has been increasing clinical interest in biologically driven, minimally invasive regenerative alternatives aimed at optimizing bone healing through enhanced angiogenesis, osteogenesis, and modulated inflammatory responses. Two prominent candidates within these regenerative strategies include Simvastatin and Platelet-Rich Fibrin (PRF).
Simvastatin, primarily known for its lipid-lowering properties, has been documented extensively for its osteogenic potential in bone regeneration. It acts by inhibiting the mevalonate pathway, thereby stimulating osteoblastic differentiation, enhancing bone morphogenetic protein-2 (BMP-2) expression, and promoting angiogenesis through increased vascular endothelial growth factor (VEGF) production. Moreover, Simvastatin demonstrates significant anti-inflammatory properties, making it a favorable adjunctive therapy for post-extraction socket management [7]. Its localized delivery via carriers such as gel foam provides targeted therapeutic action with minimal systemic exposure, thereby reducing potential adverse reactions and improving patient tolerance.
On the other hand, Platelet-Rich Fibrin (PRF), a second-generation autologous platelet concentrate, represents a widely explored biological regenerative material. PRF consists of a fibrin matrix embedded with activated platelets, leukocytes, and concentrated growth factors, including platelet-derived growth factor (PDGF), transforming growth factor-beta (TGF-β), and VEGF. These constituents collectively facilitate bone healing by promoting cellular proliferation, angiogenesis, and collagen deposition, while concurrently providing a natural biological scaffold that stabilizes the extraction socket and enhances soft-tissue healing. Its autologous nature significantly reduces the risk of immunogenic reactions, thereby ensuring optimal biocompatibility and minimal complications [7-9].
Despite these promising individual properties, comparative studies explicitly assessing the regenerative effectiveness of Simvastatin and PRF within mandibular molar extraction sites remain limited. The mandible, characterized by denser cortical bone, distinctive vascular architecture, and higher occlusal loading, presents unique challenges that necessitate targeted investigation. A comprehensive understanding of the comparative efficacy of these two biologically oriented approaches in this anatomical region would substantially contribute to refining clinical decision-making, optimizing therapeutic protocols, and achieving superior clinical outcomes. Thus, this study aimed to investigate and compare the regenerative and healing capacities of Simvastatin and PRF post-mandibular molar extraction.
2. MATERIALS AND METHODS
2.1. Study Design and Ethical Considerations
This prospective, randomized, parallel-arm trial enrolled 40 patients who required mandibular molar extraction from March 2023 to September 2024. Ethical clearance was obtained from the Institutional Ethical Committee of Datta Meghe Institute of Medical Sciences, adhering strictly to the Helsinki Declaration. The IRB approval Ref. No. DMIHER (DU)/IEC/2023/842 was assigned to the manuscript.
2.2. Sample Size Calculation
Sample size was determined using a standard statistical formula based on anticipated differences in bone regeneration outcomes. With significance (α=0.05) and power (80%), the minimum sample required per group was calculated as 20, totaling 40 participants.
2.3. Patient Selection
Inclusion criteria included systemically healthy patients (ASA I) aged 16–50 years undergoing mandibular molar extraction. Exclusion criteria involved systemic diseases, immunocompromised status, pregnancy, smoking, alcoholism, bisphosphonate therapy, and local pathology like cysts or tumors.
2.4. Randomization and Interventions
Participants were randomly allocated to two groups via computer-generated randomization:
- Group A (Simvastatin): Simvastatin (10mg) impregnated gel foam placed in extraction sockets.
- Group B (PRF): Autologous PRF prepared from 10 ml centrifuged blood samples placed into the extraction sockets.
Procedures followed standardized surgical protocols performed by a single surgeon. Postoperatively, analgesics and antibiotics were prescribed uniformly.
2.5. Outcome Measures
Outcomes included postoperative pain (VAS scores on days 1, 3, 7), soft tissue healing (Landry’s scale on days 1, 3, 7), bone density (CBCT measured at baseline and 3 months), and postoperative complications.
2.6. Statistical Analysis
Analyses were anticipated with 3-month CBCT bone density (HU) as the primary endpoint and pain (VAS day 1/3/7), soft-tissue healing (Landry day 1/3/7), extraction time, and postoperative complications as endpoints anticipated for secondary analyses. Two-sided testing (α=0.05) was used under an intention-to-treat approach; data are given as mean±SD or n (%). Comparability of baseline (age, sex, duration) was tested using independent t tests and χ2/Fisher's exact tests. Group differences for single–time–point data (e.g., 3-month HU) were tested using independent t tests (95% CIs) or Mann–Whitney tests (when nonparametric). Repeated measures (VAS, Landry, immediate vs 3-month HU) were controlled with linear mixed models including fixed effects for (group, time, and group×time), and random participant intercept. Complication rates were compared using Fisher's exact test and presented as risk ratios (95% CIs). Missing data ≤5% were imputed using complete-case analysis; for missing data >5%, multiple imputation (m=20) was used. Secondary endpoints were exploratory with Benjamini–Hochberg FDR adjustment (q=0.10). Statistical analyses were performed in R (v4.3).
3. RESULTS
Figure 1 represents the CONSORT flowchart for this study.
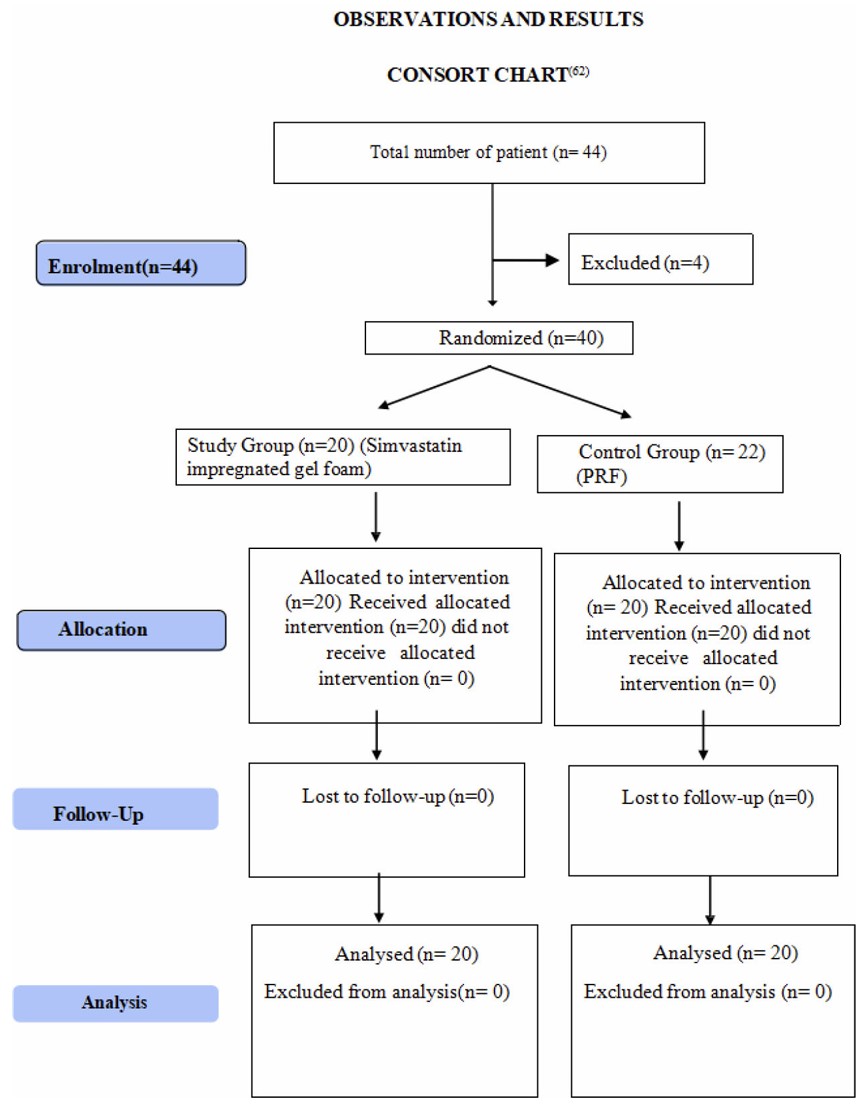
Process of randomization for the study as per the CONSORT guidelines.
Demographic data showed no significant differences in mean age (Simvastatin: 34.2±6.8 years, PRF: 33.9±7.1 years; p =0.82) or gender ratio (60% male, 40% female in both groups) (Table 1).
| Group | Age (mean±SD) | Gender Ratio (M:F) | Duration of Extraction (mean±SD min) |
|---|---|---|---|
| Simvastatin | 34.2±6.8 | 12:8 | 15.4±3.2 |
| PRF | 33.9±7.1 | 12:8 | 15.1±2.9 |
VAS scores decreased from day 1 to day 7, with no significant intergroup differences at any point (Table 2). Soft tissue healing also improved progressively with no significant differences (p >0.05). Bone density was significantly higher in the Simvastatin group (422±30 HU) compared to PRF (411±28 HU; p =0.01) at 3 months. Complications were minimal, with only one dry socket and one infection reported in the PRF group.
| Outcome Measure | Time Point | Simvastatin (mean±SD) | PRF (mean±SD) | p-value |
|---|---|---|---|---|
| VAS Score | Day 1 | 4.8±0.9 | 5.1±1.2 | 0.18 |
| Day 7 | 1.5±0.5 | 1.6±0.6 | 0.12 | |
| Bone Density | 3 months (HU) | 422±30 | 411±28 | 0.01 |
The extraction duration, as shown in Table 3 and Figure 2, was comparable between the groups, with the Simvastatin group having a mean extraction duration of 15.4 ± 3.2 minutes and the PRF group 15.1 ± 2.9 minutes. The p-value of 0.67 suggested that there was no significant difference in the time required for the extraction procedure between the two groups. This further indicated that external surgical factors were standardized across the groups, minimizing procedural variability.
| Group | N | Mean | Std. Deviation | Std. Error Mean | t-value | p-value |
|---|---|---|---|---|---|---|
| Simvastatin | 20 | 15.4 | 3.2 | 0.68 | 0.43 | 0.67 |
| PRF | 20 | 15.1 | 2.9 | 0.63 |
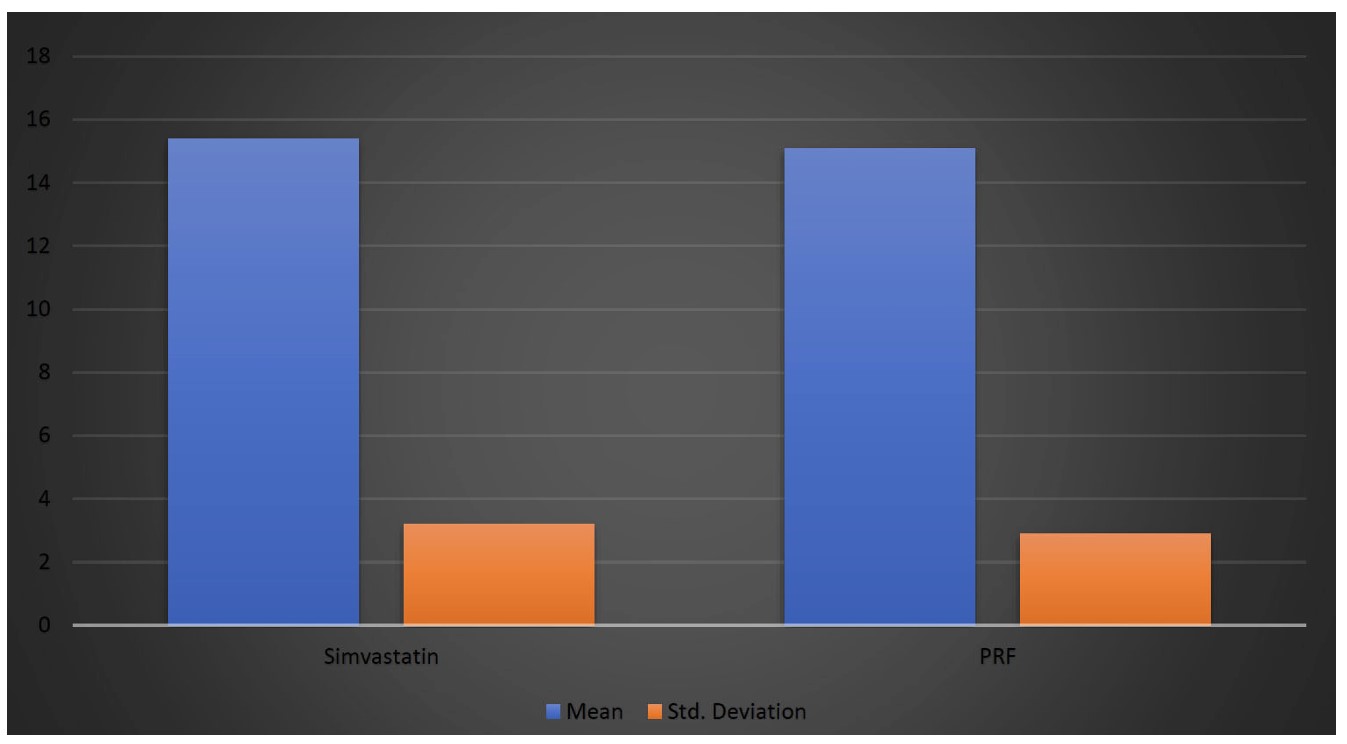
Graphical representation of the duration of extraction in the two groups.
Pain levels, assessed using the Visual Analog Scale (VAS), decreased in both groups (Table 4 and Figure 3, respectively). On Day 1 postoperatively, the mean VAS score was 4.8 ± 0.9 in the Simvastatin group and 5.1 ± 1.2 in the PRF group (p = 0.18). By Day 3, the pain scores had decreased in both groups, with the Simvastatin group recording 3.2 ± 0.8 and the PRF group 3.6 ± 1.0 (p = 0.09). On Day 7, both groups showed further pain reduction, with mean scores of 1.5 ± 0.5 in the Simvastatin group and 1.6 ± 0.6 in the PRF group. The difference at Day 7 was not statistically significant (p = 0.12), indicating comparable pain relief outcomes between the two interventions during the postoperative period.
| Time Point | Simvastatin Group (mean ± SD) | PRF Group (mean ± SD) | p-value |
|---|---|---|---|
| Day 1 Post-op | 4.8 ± 0.9 | 5.1 ± 1.2 | 0.18 |
| Day 3 Post-op | 3.2 ± 0.8 | 3.6 ± 1.0 | 0.09 |
| Day 7 Post-op | 1.5 ± 0.5 | 1.6 ± 0.6 | 0.12 |
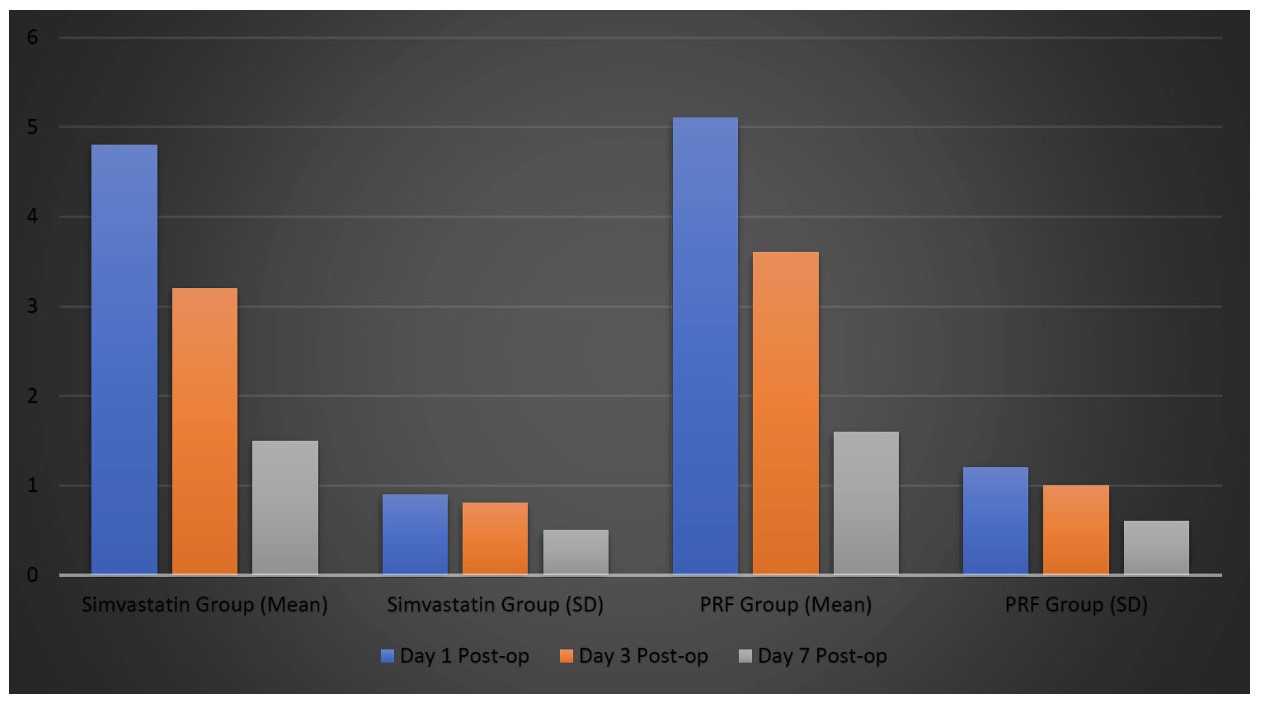
Graphical representation of pain scores on visual analog scale (VAS).
The healing outcomes showed a progressive improvement in both soft tissue and bone healing over time in both groups (Table 5 and Figure 4, respectively). On Day 1, the soft-tissue healing scores were slightly higher in the Simvastatin group (2.8) than in the PRF group (2.5), though the difference was not statistically significant (p = 0.21). By Day 3, the scores had improved further, with the Simvastatin group reaching 4.1 and the PRF group 3.8 (p = 0.18), indicating a slightly faster healing rate in the Simvastatin-treated patients. By Day 7, soft-tissue healing was nearly complete, with scores of 4.7 and 4.5 in the Simvastatin and PRF groups, respectively, suggesting no significant difference in final healing outcomes.
| Time Point | Soft Tissue Healing Score as per Landry’s Scale (1-5) - Simvastatin Group | Soft Tissue Healing Score as per Landry’s Scale (1-5) - PRF Group | p-value |
|---|---|---|---|
| Day 1 | 2.8 | 2.5 | 0.21 |
| Day 3 | 4.1 | 3.8 | 0.18 |
| Day 7 | 4.7 | 4.5 | 0.12 |
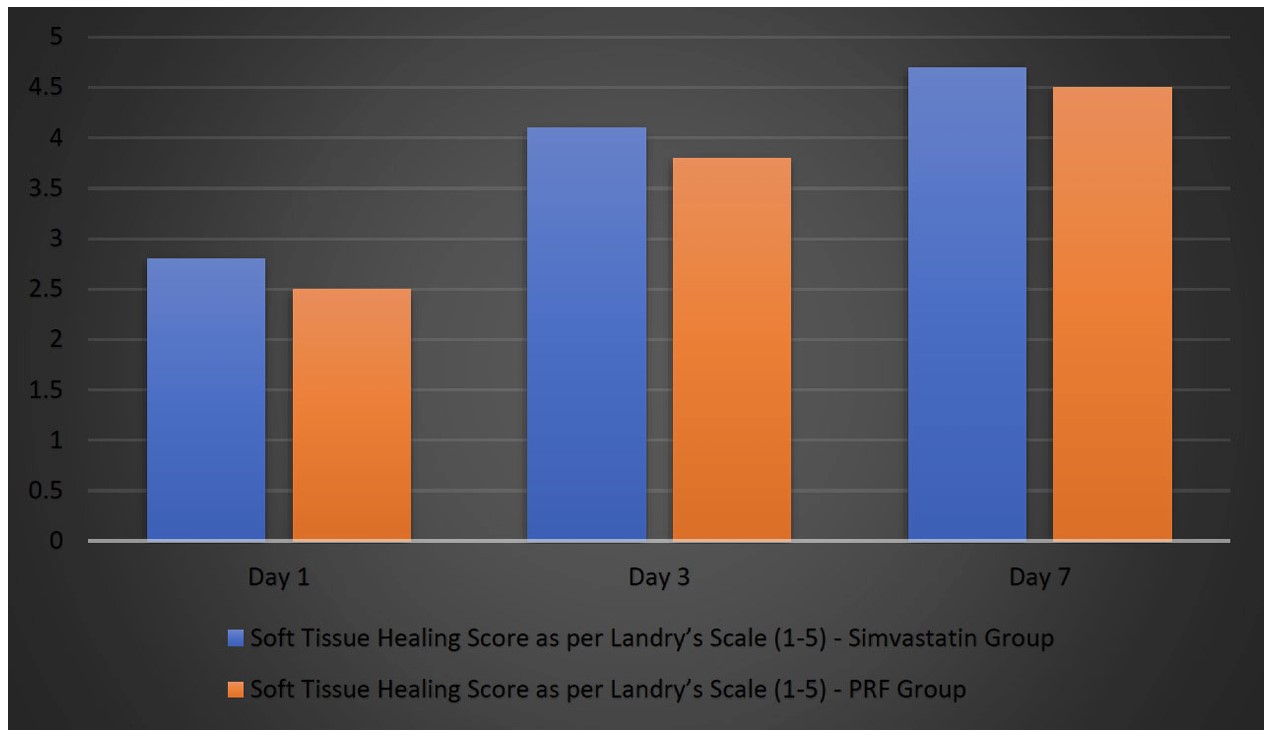
Graphical representation of healing outcomes.
Bone density, measured by CBCT, significantly improved for both groups over time (Table 6 and Figure 5, respectively). Immediately postoperatively, bone density values were comparable between the two groups, with the Simvastatin group recording a mean density of 312 ± 22 HU, and the PRF group recording a slightly lower mean of 305 ± 25 HU. This difference was not statistically significant (p = 0.32), suggesting that both interventions exhibited similar initial bone density. At the 3-month postoperative mark, an important difference emerged between the groups. The Simvastatin group demonstrated a mean bone density of 422 ± 30 HU, whereas the PRF group exhibited a slightly lower value of 411 ± 28 HU. This difference was statistically significant (p = 0.01), indicating that Simvastatin supplementation led to greater bone regeneration than PRF during this period. This suggests that though both treatments supported bone regeneration, the results in the Simvastatin group were superior at 3 months after the surgery.
| Time Point | Simvastatin Group (mean ± SD) | PRF Group (mean ± SD) | p-value |
|---|---|---|---|
| Immediate Post-op | 312 ± 22 | 305 ± 25 | 0.32 |
| 3 months Post-op | 422 ± 30 | 411 ± 28 | 0.01 |
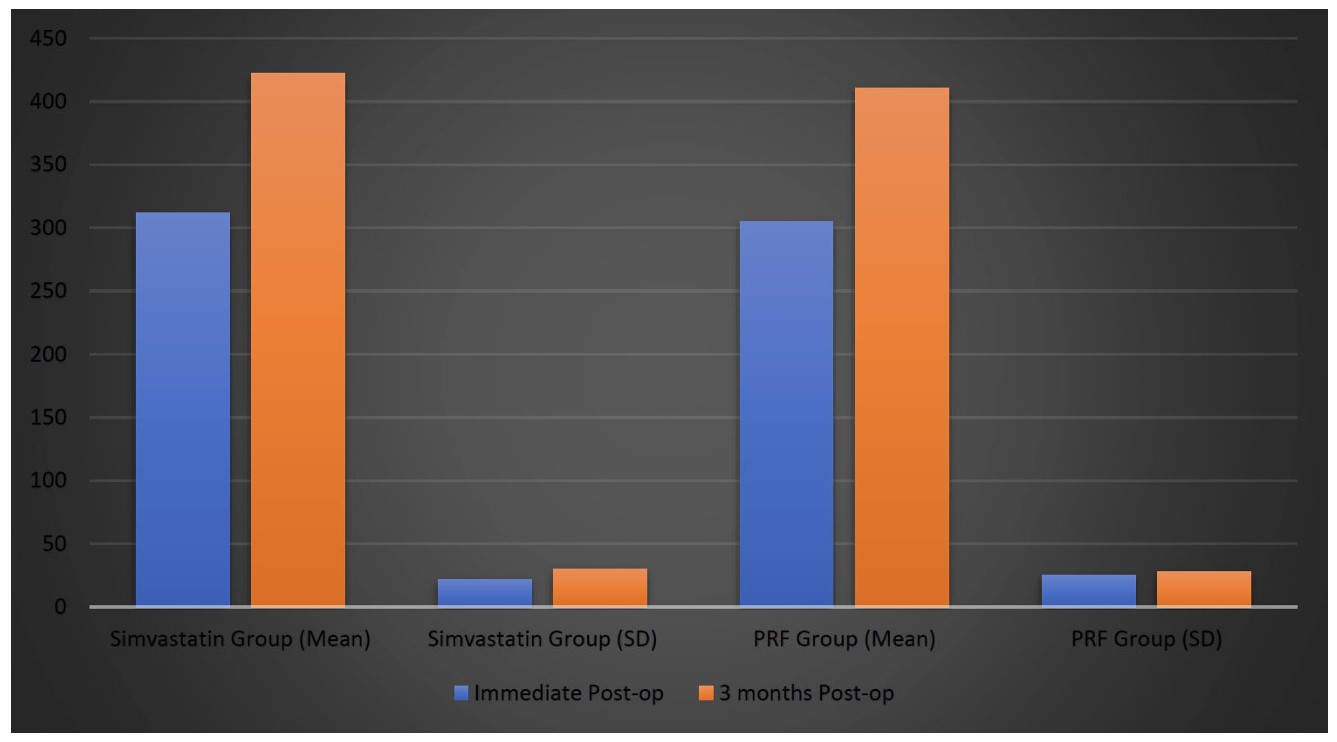
Graphical representation of bone regeneration (bone density measured by CBCT in HU).
The rate of postoperative complications was low and did not differ between the two groups (Table 7 and Figure 6, respectively). Dry socket occurred in 1 patient (5.0%) in the PRF group and none in the Simvastatin group, but this difference was not statistically significant (p = 0.50). Similarly, infection was noted in 1 patient (5.0%) in the PRF group, while none were reported in the Simvastatin group; again, this difference was not statistically significant (p = 0.50). There were no incidences of secondary bleeding or the need for re-suturing in either group, resulting in no statistically significant difference (p = 1.00 for both outcomes).
| Complication | Simvastatin Group (n, %) | PRF Group (n, %) | p-value |
|---|---|---|---|
| Dry Socket | 0 (0.0%) | 1 (5.0%) | 0.50 |
| Secondary Bleeding | 0 (0.0%) | 0 (0.0%) | 1.00 |
| Infection | 0 (0.0%) | 1 (5.0%) | 0.50 |
| Need for Re-suturing | 0 (0.0%) | 0 (0.0%) | 1.00 |
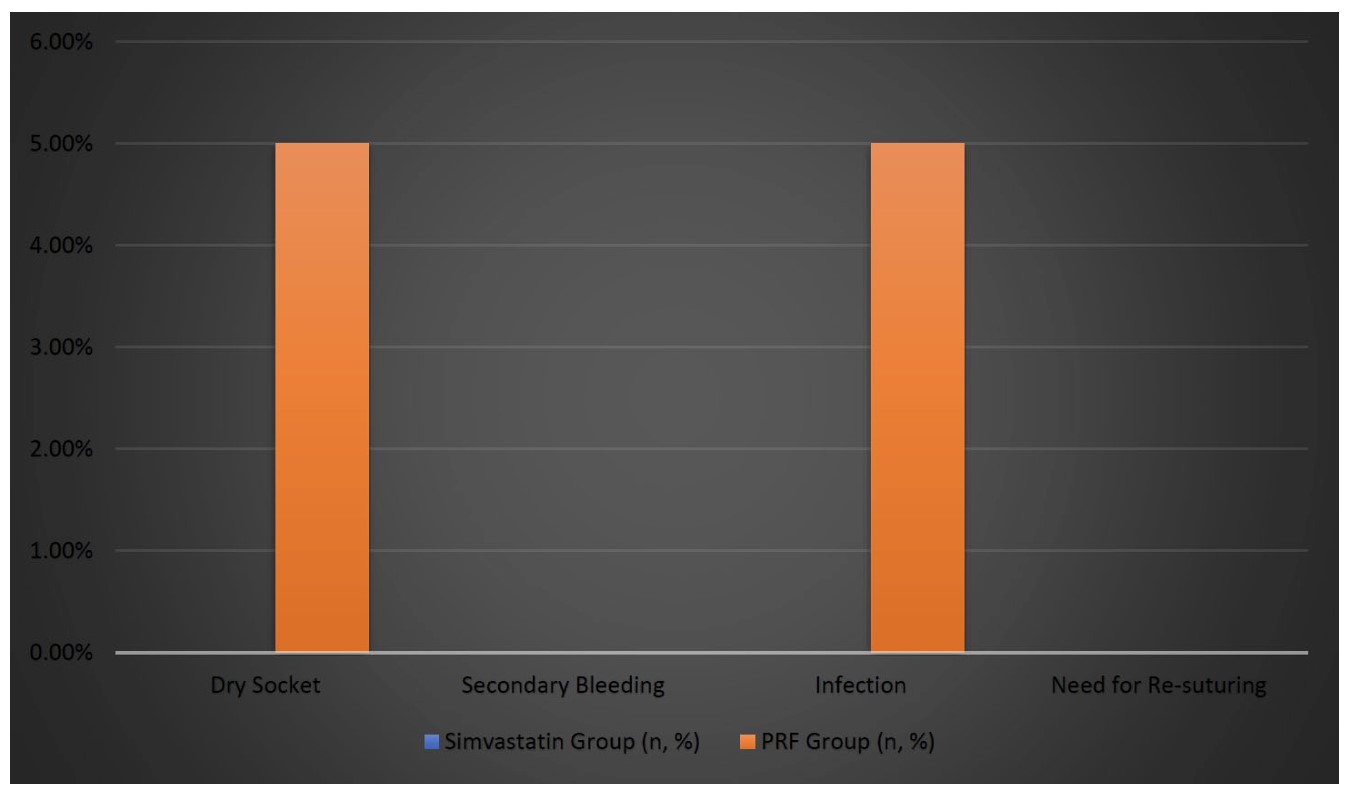
Graphical representation of postoperative complications.
4. DISCUSSION
Alveolar ridge preservation following mandibular molar extraction has profound implications for clinical outcomes in prosthetic rehabilitation, implant placement, and long-term periodontal health [10-12]. The present study provided a direct comparative analysis between two biologically-oriented regenerative techniques: Simvastatin-impregnated gel foam and Platelet-Rich Fibrin (PRF). Both interventions demonstrated encouraging results in postoperative pain management, soft tissue healing, and bone regeneration; however, the Simvastatin group showed distinct advantages, particularly enhanced bone regeneration at the three-month evaluation.
Consistent with the literature, the present study established that postoperative pain levels, assessed using the Visual Analog Scale (VAS), gradually declined over the first postoperative week without statistically significant differences between the Simvastatin and PRF groups. Previous studies have reported that Simvastatin exerts anti-inflammatory effects by inhibiting inflammatory cytokines such as IL-1β and TNF-α, thereby contributing to postoperative pain reduction and improved patient comfort [13, 14]. The lack of significant differences observed in the current trial could be attributed to both materials’ inherent anti-inflammatory properties, underscoring their suitability for post-extraction applications.
Regarding soft-tissue healing, both treatment groups showed progressive improvement over the postoperative period, as assessed by Landry’s healing index. Although no statistically significant differences emerged, Simvastatin-treated sockets consistently showed marginally superior healing outcomes. This finding aligns with prior research, which emphasizes Simvastatin’s potential role in promoting epithelialization and collagen synthesis, potentially via enhanced angiogenesis and modulated local inflammatory responses [15-17]. Conversely, PRF also contributed positively by sustained delivery of growth factors such as PDGF and TGF-β, fostering angiogenesis and collagen deposition; however, its comparatively gradual and prolonged mode of action may explain the minor differences observed between groups [18, 19].
The critical finding of this investigation was the significantly enhanced bone regeneration in sockets treated with Simvastatin at three months postoperatively. CBCT assessments revealed higher mean bone density in Simvastatin-treated sites (422 ± 30 HU) versus PRF-treated sites (411 ± 28 HU), with statistical significance (p =0.01). The pronounced osteogenic effect of Simvastatin may be primarily attributed to its potent ability to enhance osteoblastic differentiation via BMP-2 induction, facilitate angiogenesis through VEGF upregulation, and mitigate bone resorption by modulating the RANKL/OPG pathway [14, 20]. PRF, while promoting effective bone healing through growth factor-mediated mechanisms, typically achieves regeneration over a more extended period, potentially explaining the modestly lower bone density observed at three months [19-21].
Complication rates were minimal across both groups, underscoring the clinical safety of both materials. However, isolated cases of dry socket and infection were observed only in the PRF group, albeit without statistical significance. This finding suggests that Simvastatin’s additional antimicrobial properties and robust anti-inflammatory effects may offer a marginal clinical advantage in reducing postoperative complications [13, 16].
Given the present study's findings, Simvastatin-impregnated gel foam emerges as an effective, minimally invasive alternative for alveolar ridge preservation, providing superior bone regenerative outcomes compared to PRF. However, the current study’s limitations—including a relatively small sample size, short-term follow-up, and lack of advanced diagnostic evaluations—highlight the need for further large-scale clinical investigations. Future studies with longer follow-up periods, multicentric designs, histological analyses, and molecular profiling would enhance our understanding of the underlying regenerative mechanisms and the clinical efficacy of these biologically driven preservation strategies.
5. LIMITATIONS
This study’s limitations include its small sample size, short follow-up duration, and the absence of advanced diagnostic analyses (histological and molecular). Patient-specific factors such as systemic health and smoking status were not extensively explored. The single-center design may introduce operator bias. Larger, multicenter studies are recommended.
CONCLUSION
This study compared the efficacy of Simvastatin-impregnated gel foam and Platelet-Rich Fibrin (PRF) in promoting postoperative healing, pain relief, and bone regeneration following mandibular molar extractions. Baseline characteristics, including age, gender, and surgical duration, were comparable between groups. Postoperative pain scores (VAS) decreased progressively in both groups without significant differences. Soft tissue healing assessed by Landry’s scale was similar, with slightly faster recovery observed in the Simvastatin group. Cone-beam CT (CBCT) analysis at three months revealed significantly higher bone density in the Simvastatin group. Postoperative complications were minimal in both groups, with a marginally lower incidence in the Simvastatin group. Overall, both Simvastatin and PRF effectively supported alveolar ridge preservation post-extraction, with Simvastatin providing superior bone regenerative outcomes. These findings suggest simvastatin may be advantageous for accelerated regeneration scenarios.
AUTHORS’ CONTRIBUTIONS
The authors confirm contribution to the paper as follows: S.K. and N.B.: Study conception and design were carried out; D.S.: Data collection was performed; S.K., D.S., and N.B.: Analysis and interpretation of the results involved; S.K. and D.S.: The draft manuscript was prepared. All authors reviewed the results, revised the manuscript, and approved the final version.
LIST OF ABBREVIATIONS
| IOD | = Implant overdenture |
| 1-IOD | = Single implant overdenture |
| MIOD | = Multiple implant–supported overdenture |
| PROMs | = Patient-reported outcome measures |
| RCT | = Randomized controlled trial |
| CI | = Confidence interval |
| PRISMA | = Preferred Reporting Items for Systematic Reviews and Meta-Analyses |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Ethical clearance was obtained from the Institutional Ethical Committee of Datta Meghe Institute of Medical Sciences (IRB approval Ref. No. DMIHER (DU)/IEC/2023/842).
HUMAN AND ANIMAL RIGHTS
All procedures performed in studies involving human participants were in accordance with the ethical standards of institutional and/or research committee and with the 1975 Declaration of Helsinki, as revised in 2013.
CONSENT FOR PUBLICATION
Written informed consent was obtained from all participants prior to enrollment.
AVAILABILITY OF DATA AND MATERIALS
All data generated or analyzed during this study are included in this published article.
ACKNOWLEDGEMENTS
Declared none.


