All published articles of this journal are available on ScienceDirect.
Microtensile Bond Strength and Microhardness of Resin–dentin Bonds using Different Resin-based Composite Systems
Abstract
Introduction
Dental caries remain a major global health concern, emphasizing the need for durable restorative materials with reliable mechanical properties. This study aimed to compare the μTBS and Vickers hardness number (VHN) of three composite materials (3M Filtek Z250, Kerr OptiShade, and SHOFU Beautifil II LS) on sound coronal dentin using a corresponding bonding system (3M Universal Bond, Kerr OptiBond, and SHOFU BeautiBond Universal Adhesive, respectively).
Methods
Dentin surfaces were exposed from twelve extracted molars. Each composite and its respective adhesive were applied and cured. The teeth were cut into 1 × 1 × 8 mm beams. Twenty beams per group were tested for μTBS. Additionally, 20 composite discs (2 x 10 mm) were prepared for each material. VHN was measured at three sites per disc, and mean values were calculated.
Results
Filtek Z250 showed the highest VHN, followed by OptiShade and Beautifil II LS, with significant differences among all groups. OptiShade demonstrated the highest μTBS, followed by Filtek Z250 and Beautifil II LS. μTBS differed significantly between Beautifil II LS and both Filtek Z250 and OptiShade but not between Filtek Z250 and OptiShade.
Discussion
Differences in filler composition, resin matrix characteristics, and adhesive protocols likely contributed to the variations observed in microhardness, bonding performance, and restoration longevity.
Conclusion
Filtek Z250 exhibited superior microhardness, while OptiShade demonstrated superior bond strength. Beautifil II LS performed the worst in both tests. These findings support evidence-based material selection to enhance restoration longevity.
1. INTRODUCTION
Dental caries represent a rapidly growing global public health concern, affecting 3.5 billion individuals worldwide [1]. Given the number of individuals seeking dental care, it is vital that direct restorative materials be durable, fracture-resistant, and exhibit excellent mechanical, physical, and thermal properties [2-4]. Many dental biomaterials have been developed to formulate an ideal restorative material that meets patients' clinical needs; however, none have met all of the aforementioned properties, and resin-dentin bond stability remains a challenge [5-7].
While resin composites are currently the most used restorative material, several factors can affect their physical and mechanical properties [2]. According to the Academy of Dental Materials’ criteria, resin composites are evaluated by measuring their strength, elastic modulus, toughness, fatigue resistance, hardness, and wear [2]. Most dental restorations fail due to tensile stresses within the material’s structure due to their complex geometries. Therefore, tensile strength is one of the most important properties to be measured when developing a standardized dental material [2].
Microtensile bond strength (μTBS) testing is a crucial aspect of restorative dentistry. It evaluates the adhesive performance of dental materials, specifically between the resin-based composites and adhesives used in the bonding of restorations to tooth structures [2]. By subjecting the materials to tensile forces, mirroring the stresses encountered during mastication and various functional movements in the oral environment, the μTBS test provides valuable insights into the material’s ability to adhere to the tooth structure [8]. By simulating real-world conditions, this comprehensive assessment enables a more localized assessment of bond strength, allowing researchers and practitioners to identify weak points or areas susceptible to adhesive failure [9]. Recognizing and comprehending the intricacies of bond strength is crucial for predicting the longevity and success of restorations. This knowledge also helps clinicians select materials and employ techniques that can effectively withstand the challenges of the oral cavity [2].
Hardness is a surface mechanical property that is defined as the resistance of the material to permanent indentation [10]. Hardness tests have been utilized to study and characterize resin composites for more than a century due to their ease of use [2]. Hardness is a critical property because it directly correlates with a material’s ability to resist wear, withstand occlusal forces, and maintain structural integrity within the challenging confines of the oral environment. To achieve an acceptable degree of polymerization according to ISO standards, a bottom-to-top microhardness ratio of at least 80% must be achieved at a depth of 2 mm [11]. Therefore, the composite material must exhibit not only hardness but also durability to provide a long-lasting restoration that withstands the daily wear and tear of normal use [2].
Resin composites exhibit numerous advantageous properties compared to other restorative materials; however, they also have some drawbacks, such as a lack of antibacterial or remineralization effect, which play an important role in caries formation [12]. Their other drawbacks include, but are not limited to, polymerization shrinkage, microleakage, secondary caries, marginal staining, debonding of the restoration, postoperative pain and sensitivity, cusp deflection, and restoration fracture [12].
Kerr (Brea, CA, USA) introduced OptiShade composites to the market, which have adaptive responsive technology [10]. This technology makes it easier for this composite material to mimic natural teeth due to its nanoparticle fillers. It also reinforces structural integrity with spherical zirconia and silica particles, which leads to a more effective polymerization process and increased strength, thereby providing the restorations with outstanding esthetics and strength [10]. This composite material is easily polishable, has dynamic viscosity, and reflects light similarly to enamel [10].
To minimize these outcomes, low-shrinkage composites such as Beautifil II LS (SHOFU, San Marcos, CA, USA) have been developed [13]. The low shrinkage of the Beautiful II LS composite aims to minimize potential microleakage and postoperative sensitivity [14]. SHOFU claims that the Beautiful II LS composite also has an anti-plaque effect and fluoride-releasing capacity, offering patients effective protection against secondary caries [14]. The hardness of the glass filler used as raw material in the Beautifil II LS composite is similar to that of enamel, which increases the composite’s overall strength [14]. However, the stability of the resin–dentin bond in this composite has not yet been thoroughly investigated.
It could be hypothesized that adhesive dentistry aims to accomplish a close, long-lasting bond between the restorative material and the dental structures [15]. One-step bonding techniques have been preferred in dental practice due to their ease of use and efficient reduction of procedure time [16]. Kerr developed OptiBond, a hydrophilic solution meticulously crafted to align with the natural tooth structure’s composition [17]. This innovative technology ensures excellent penetration into dentinal tubules, thereby protecting against potential issues such as microleakage and postoperative sensitivity [17]. The OptiBond formulation, enriched with the glycerophosphate dimethacrylate monomer-considered the gold standard in adhesion-represents a breakthrough in achieving unparalleled compatibility with both the tooth surface and the resin material [17], thereby contributing to greater bond strength and emphasizing OptiBond's efficacy [17].
In comparison, SHOFU’s BeautiBond Universal Adhesive exhibits a high bond strength to natural teeth due to its 2-hydroxyethyl methacrylate-free composition [16]. The BeautiBond Universal Adhesive has been compared with a two-step adhesive (SHOFU’s FL-Bond II) and has shown similar shear bond strength after 24 hours [16]. To provide greater insight into the stability of the resin–dentin bond, μTBS tests estimate the level of stress required to initiate debonding [18].
In conclusion, hardness is a crucial characteristic because it is clearly linked to a material’s ability to withstand wear, withstand occlusal forces, and maintain its structural integrity under the demanding conditions of the oral environment. Given the complexities of dental materials, μTBS tests are a pivotal tool, providing valuable insights into adhesive performance and aiding in identifying weak points. Overall, this comprehensive understanding contributes to selecting materials and techniques that can withstand the multifaceted challenges of the oral environment, ensuring the long-term success of restorative procedures.
Therefore, this study compares the μTBS and Vickers hardness number (VHN) on sound coronal dentin of a novel resin composite (Beautifil II LS) bonded with BeautiBond Universal Adhesive with two of the most commonly used resin composites in Saudi Arabia: Filtek Z250 bonded with Universal Bond (3M, St. Paul, MN, USA) and OptiShade bonded with OptiBond.
2. MATERIALS AND METHODS
This in vitro study compared three resin composites bonded to sound coronal dentin on 12 non-carious molars using their corresponding bonding agent. The microtensile test was conducted with a universal testing system (Instron, Norwood, MA, USA). The microhardness test was conducted with a HMV series microhardness tester (Shimadzu, Kyoto, Japan). The experimental groups are shown in Figs. (1 and 2).
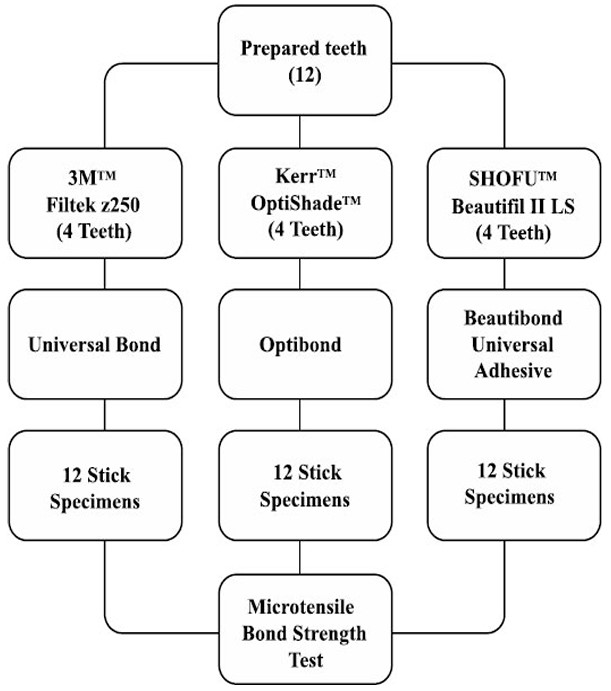
The experimental groups examined in the μTBS test.
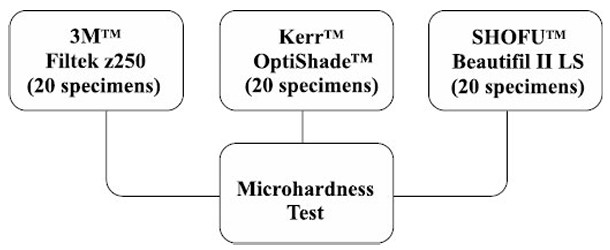
The experimental groups examined in the microhardness test.
2.1. Ethical Considerations
This study was approved by the Research Ethics Committee of the Faculty of Dentistry at King Abdulaziz University (Jeddah, Saudi Arabia; approval number: 141-07-23) and conducted in accordance with the Declaration of Helsinki. All procedures followed institutional guidelines for infection prevention and control. Informed consent was obtained from all participants prior to the use of their extracted teeth for research purposes.
2.2. µTBS Test
2.2.1. Selection
This study used 12 non-carious molars obtained from oral surgery clinics following standard clinical procedures.
2.2.2. Storage
The teeth were stored in normal saline (0.9% sodium chloride) for 1 month after extraction at a controlled room temperature of 23°C–25°C.
2.2.3. Sample Size
The required sample size was estimated using G*Power software based on a one-way analysis of variance (ANOVA) and data from Bedran-Russo et al. [19], with an effect size of 9.27, a significance level of 0.5, and a power of 80%. The power analysis indicated a sample size of 20 beams per group, for a total of 60 beams.
2.2.4. Sample Preparation
The 12 teeth were prepared using a cutting disc, removing occlusal enamel and superficial dentin, and were then randomly assigned to the three groups. The three bonding agents were applied using microbrushes and light-cured using a 3M EliparDeepCure-L LED curing light; the light intensity (1400 mW/cm2) was verified using a radiometer according to the manufacturer’s instructions. The 3M Universal Bond was applied to the dentin surface and rubbed for 20 seconds. Then, air was applied to the treated area for 5 seconds to evaporate the solvents, and the bonding agent was cured for 10 seconds. Similarly, Kerr OptiBond was applied to the dentin surface and rubbed for 20 seconds. Then, air was applied for 5 seconds, followed by curing the bonding agent for 5 seconds. The SHOFU BeautiBond Universal Adhesive was applied to the dentin surface and left undisturbed for 10 seconds. Then, it was gently dried for 3 seconds, followed by drying with strong air until a uniform layer formed. Finally, the bonding agent was cured for 5 seconds.
The resin composites were placed in 2 mm increments over their respective bonding agents. They were light-cured according to the manufacturer’s instructions at 1400 mW/cm2. The 3M Filtek Z250 and Kerr OptiShade composites were cured for 10 seconds, while the SHOFU Beautifil II LS composite was cured for 5 seconds.
The teeth were sectioned using a cutting disc to create as many beams as possible, each measuring 1 × 1 × 8 mm; those showing pulp were excluded. Then, 20 specimens from each group were mounted in the microtensile machine and subjected to the μTBS test at a crosshead speed of 0.5 mm/min.
2.2.5. Fracture Analysis
After evaluating the μTBS, each debonded specimen was examined under a stereomicroscope to determine the failure mode. The failure modes were categorized into the following five groups:
1- Between the resin composite and the bonding system.
2- Between dentin and the bonding system.
3- Within the dentin.
4- Within the composite resin.
5- Involving both the dentin/bonding system and the resin composite/bonding system.
2.3. Microhardness Test
2.3.1. Sample Preparation
For each composite material, 20 resin-based composite discs measuring ~2 mm in thickness and ~10 mm in diameter were prepared by packing the material into a mold placed over a glass slab to ensure a flat, standardized base, and curing it using an LED light source at 1400 mW/cm2 according to the manufacturer’s instructions: the Filtek Z250 and OptiShade discs for 10 seconds, and the Beautifil II LS discs for 5 seconds. The smooth surface formed in contact with the glass slab was used for microhardness testing. While no additional pressure standardization device was used, the use of a glass slab helped minimize variability in thickness and surface smoothness and reduce the risk of void formation.
The specimens were numbered from 1 to 60 and subjected to the Vickers microhardness test by a blinded researcher; the VHN values were later grouped by material. Briefly, the specimens were individually secured and placed perpendicularly to the microhardness tester’s tip. Then, a diamond indenter applied a consistent load of 980.7mN (~100 g) for 30 seconds, following standard microhardness testing protocols. The VHN value was computed automatically by the tester’s software. Three indents were created on each specimen, and the mean VHN was calculated. The specimens were tested within 24 hours of preparation and stored in a dry state during this period. The parameters were kept uniform across all specimens to ensure accurate and comparable measurements.
2.4. Statistical Analysis
The statistical analyses were conducted using SPSS Statistics (version 25.0; IBM Corp., Armonk, NY, USA). A p < 0.05 was considered statistically significant. In the Vickers microhardness analyses, the independent variable was the three different composites applied to the specimens, and the outcome variable was the VHN value. The mean and variation of the VHN values were compared between groups using the Kruskal–Wallis test and pairwise post-hoc tests. In the μTBS analyses, the independent variable was the three different composite materials, and the outcome variable was the μTBS value. The mean and variation of the μTBS values were compared between groups using one-way ANOVA and pairwise post-hoc tests.
3. RESULTS
This experimental and comparative in vitro study assessed the microhardness and shear bond strength of three composite materials. The composite materials assessed in this study are summarized in Table 1, and the mean ± standard deviation (SD) and median (interquartile range [IQR]) of their VHN values are presented in Table 2. Since the VHN data were not normally distributed (p = 0.000, Shapiro–Wilk test), they were compared between groups using non-parametric tests. The VHN values differed significantly among the three composite materials (χ2(2) = 50.694, p < 0.001; Kruskal–Wallis test), with post-hoc pairwise tests revealing significant differences between all pairs of groups (all p < 0.001, Mann–Whitney U test). Filtek Z250 had the highest VHN (greatest strength), followed by OptiShade and then Beautifil II LS.
| Material | Type | Composition | Manufacturer |
|---|---|---|---|
| Filtek Z250 | Microhybrid composite | Matrix: Bis-GMA, UDMA, and Bis-EMA. Filler: silica/zirconia. |
3M |
| OptiShade | Nano-hybrid composite | Matrix: Bis-GMA, TEGDMA, and Bis-EMA. Filler: barium glass filler, silica, ytterbium trifluoride, and prepolymerized filler. |
Kerr |
| Beautifil II LS | Giomer | Matrix: Bis-GMA and TEGDMA. Filler: aluminum oxide, silica, aluminofluoro-borosilicate glass filler, pre-reacted glass-ionomer filler, and camphoroquinone. |
SHOFU |
| Composite Material | Sample Size | Mean ± SD | Median (IQR) | Comparison | ||
|---|---|---|---|---|---|---|
| Group A | Group B | p-value | ||||
| 1- Filtek Z250 (control) | 20 | 99.69 ±7.90 | 101.55 (5.38) | 1 | 2 | <0.001 |
| 1 | 3 | <0.001 | ||||
| 2- OptiShade | 20 | 67.87 ±8.76 | 51.70 (8.29) | 2 | 3 | <0.001 |
| 3- Beautifil II LS | 20 | 50.26 ±7.08 | 68.99 (4.26) | |||
The μTBS of the three composite materials was evaluated in MPa (MPa = N / [1 mm2 cross-section]). Since the μTBS data were normally distributed (p = 0.178, Shapiro–Wilk test), they were compared between groups using parametric tests. The μTBS values differed significantly among the three composite materials (p < 0.05, one-way ANOVA), with post-hoc pairwise comparisons revealing significant differences between specific groups (Table 3). The μTBS values differed significantly between Beautifil II LS and both Filtek Z250 and OptiShade, but not between Filtek Z250 and OptiShade. Filtek Z250 exhibited the highest μTBS values (greatest strength), followed by OptiShade and then Beautifil II LS.
| Composite Material | Sample Size | Mean ± SD (MPa) | Comparison | ||
|---|---|---|---|---|---|
| Group A | Group B | p-value | |||
| 1- Filtek Z250 with Universal Bond (control) | 20 | 24.91 ± 9.07 | 1 | 2 | 1.000 |
| 1 | 3 | <0.001 | |||
| 2- OptiShade with OptiBond | 20 | 26.9 ± 7.40 | 2 | 3 | <0.001 |
| 3- Beautifil II LS with BeautiBond Universal Adhesive | 20 | 12.96 ± 7.05 | |||
After conducting the μTBS test, the failure mode of each debonded specimen was examined blindly under a stereomicroscope. The specimens were then categorized into five groups according to their failure mode. The distribution of failure modes among the debonded specimens is summarized in Table 4. Representative SEM images of the observed failure modes are presented in Figs. (3–5). A type 1 failure (Fig. 3) was characterized by adhesive separation at the composite–adhesive interface, where the composite is distinctly separated from the adhesive layer. In contrast, a type 2 failure (Fig. 4) was characterized by debonding at the adhesive–dentin interface, with the adhesive layer separating from the underlying dentin. A type 5 failure (Fig. 5) represents a more complex failure pattern involving the composite, adhesive layer, and dentin, indicating simultaneous disruption across multiple interfaces.
| Failure Mode | Number of Specimens |
|---|---|
| Between the resin composite and the bonding system | 25 |
| Between the dentin and the bonding system | 13 |
| Within the dentin | 0 |
| Within the composite resin | 0 |
| Involving both the dentin/bonding system and the resin composite/bonding system | 22 |

Representative image of a type 1 failure, showing adhesive separation at the composite–adhesive interface (1 = composite, 2 = adhesive layer).

Representative image of a type 2 failure, showing debonding at the adhesive–dentin interface (2 = adhesive layer, 3 = dentin).
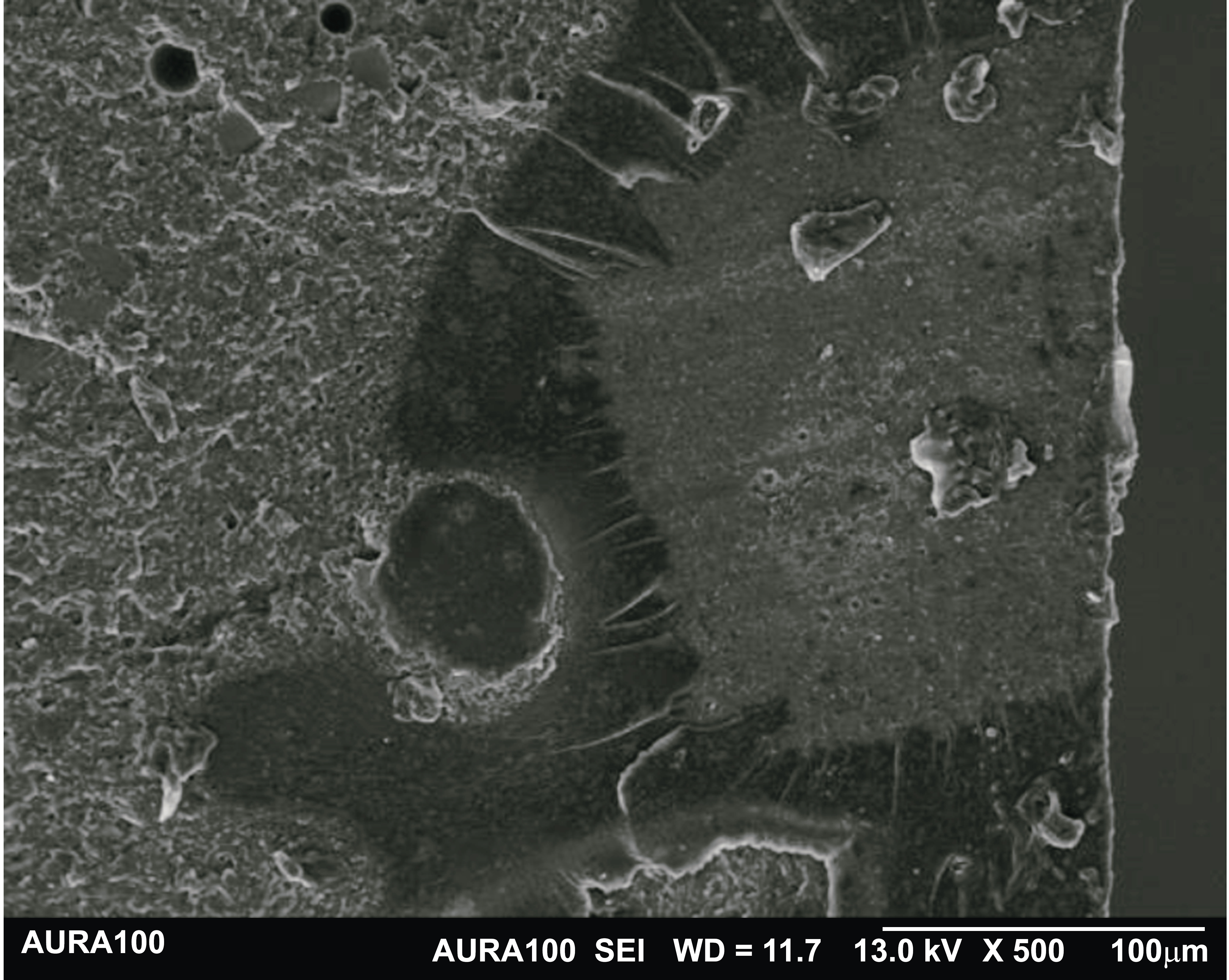
Representative image of a type 5 failure involving the composite, adhesive interface, and dentin. (1 = composite, 2 = adhesive layer, 3 = dentin).
4. DISCUSSION
The rising prevalence of dental caries globally, affecting approximately 3.5 billion individuals, underscores the need to develop advanced restorative materials in dentistry [1]. Effective resin-based composite restorations require sufficient physical, mechanical, and biological properties to withstand the abrasive conditions of the oral environment [2-4].
This study evaluated and compared the surface microhardness of the novel giomer SHOFU Beautifil II LS with two widely used composites in Saudi Arabia: 3M Filtek Z250, a microhybrid composite, and Kerr OptiShade, a nano-hybrid composite. The determined microhardness serves as an indicator of a material’s ability to withstand the abrasive and compressive forces encountered within the oral cavity. The VHN values of dental composites vary from 40 to above 100 [20]. However, to match natural teeth, a VHN of at least 50 is required [21]. Microhardness is also affected by factors such as filler type, morphology, and size; increasing filler content increases hardness.
This study assessed the microhardness of the top surface of three composite materials. Composite resin discs measuring 2mm in thickness were prepared to ensure maximum polymerization. Appropriate polymerization is critical to restoration success, as incomplete curing can result in water absorption, reduced wear resistance and strength, and the release of residual monomers [22].
Based on their composition, the Beautifil II LS, OptiShade, and Filtek Z250 composites exhibit differences in filler content, filler size, and resin matrix composition, all of which influence their strength. While the Beautifil II LS and OptiShade composites may offer advantages in terms of filler technology and resin matrix design, our results highlight notable disparities in material strength among the three composites examined, as evidenced by their significant differences in VHN values. Among the examined composites, Filtek Z250 demonstrated the greatest strength, followed by OptiShade and then Beautifil II LS. Given its relatively lower microhardness, the Beautifil II LS composite may be unsuitable for stress-bearing restorations; however, it could be considered for use in low-load or non-stress-bearing areas where its aesthetic and fluoride-releasing properties are advantageous.
Both the OptiShade and Beautifil II LS composites contain triethylene glycol dimethacrylate (TEGDMA) in their matrix. Studies have shown that composites with low TEGDMA content in their matrix exhibit lower hardness [23], which may help explain the comparatively lower VHN values observed for both the OptiShade and Beautifil II LS composites. In addition, a higher matrix-to-filler ratio generally results in a softer material, as the resin matrix is inherently less rigid than filler particles, which may also help explain the lower VHN values observed for both the OptiShade and Beautifil II LS composites. However, the specific filler content percentages for these two materials are not disclosed by their manufacturers, limiting direct comparison with Filtek Z250, which contains approximately 60% filler by volume (82% by weight). Filtek Z250 is a microhybrid composite with a larger filler size than OptiShade, a nanohybrid composite. Although nanohybrid composites are stronger, and the fillers-barium glass filler and prepolymerized filler-increase strength, Filtek Z250 exhibited greater strength than both the Beautifil II LS and OptiShade composites. Beautifil II LS is a giomer that contains glass ionomer, which also reduces its microhardness. A previous study also showed that Filtek Z250 has high microhardness not only at the surface but also at various depths, supporting its use in bulk-fill restorations [24].
This study also evaluated the μTBS of Filtek Z250, a microhybrid composite, OptiShade, a nano-hybrid composite, and Beautifil II LS, a giomer, on sound coronal dentin using their corresponding adhesives (3M Universal Bond, Kerr OptiBond, and SHOFU BeautiBond Universal Adhesive, respectively). Our analyses revealed notable differences in μTBS among the three composites examined. While the μTBS values differed significantly between the Beautifil II LS composite and both the Filtek Z250 and OptiShade composites, they did not differ significantly between the Filtek Z250 and OptiShade composites. The OptiShade composite exhibited the highest μTBS values, followed closely by the Filtek Z250 composite and then the Beautifil II LS composite.
How the dental adhesive is applied plays a crucial role in its performance [25]. The instructions for both the 3M Universal Bond and Kerr OptiBond adhesives included rubbing the bond before curing it. Rubbing universal adhesives has been shown to enhance bond strength [26], which is consistent with our results. These findings have implications for clinical practice, as the ability of dental composites to form strong bonds with tooth structure is essential to the longevity and success of dental restorations. The superior performance of OptiShade and Filtek Z250 composites in terms of μTBS suggests that they may offer more reliable adhesion and durability in clinical settings than Beautifil II LS.
The μTBS test is a crucial method for assessing the adhesive properties of materials used in dental or biomedical applications. All debonded specimens were examined under a stereomicroscope to determine their failure mode, allowing categorization into distinct groups and providing valuable insights into the performance and reliability of the adhesive systems examined. Identifying the predominant failure modes and their underlying mechanisms can help to refine adhesive formulations, optimize bonding protocols, and ultimately enhance the durability and efficacy of adhesive interfaces in various biomedical and dental applications.
Of the 60 specimens examined in our study, 25 failed between the resin composite and the bonding system, 13 between the dentin and the bonding system, and 25 combined failures involving both the dentin/bonding system and the resin composite/bonding system. The absence of failures within the dentin and the composite resin signifies satisfactory cohesive integrity.
The detachment at the interface between the resin composite and the bonding system suggests potential issues in bonding strength or compatibility, emphasizing the importance of optimizing the adhesive formulation or application technique. Failure at the dentin-bonding interface indicates challenges in achieving strong bonding to the tooth substrate, emphasizing the importance of surface treatment. Mixed failures underscore challenges in achieving uniform bonding across both substrates, suggesting the need to optimize adhesive formulation, surface treatment, and bonding techniques to enhance overall bond strength and durability.
5. LIMITATIONS
Our study was conducted in a laboratory setting, which may not fully replicate the dynamic and complex conditions of the actual oral cavity. To address this limitation, future studies could explore the application of these findings in a clinical trial setting, measuring other material properties such as dimensional stability and water absorption. Our study also did not include thermocycling to simulate long-term aging, which may lead to overestimated results.
CONCLUSION
Our findings revealed significant differences in microhardness, with the Filtek Z250 composite exhibiting superior hardness, supporting its suitability for stress-bearing areas where resistance to deformation and wear is essential, followed by the OptiShade composite and then the Beautifil II LS composite. In contrast, the OptiShade composite exhibited the highest bond strength, indicating a strong, durable adhesive interface, followed by the Filtek Z250 composite and then the Beautifil II LS composite. Although the Beautifil II LS composite exhibited weaker mechanical properties than the other two composites examined, it is a viable option for non-stress-bearing areas, such as Class III and Class V restorations, where its fluoride-releasing potential may provide additional preventive benefits. Our study contributes to improving the longevity and success of dental restorations by providing evidence-based guidance on composite resin selection based on clinical indication and stress environment.
AUTHORS’ CONTRIBUTIONS
The authors confirm contribution to the paper as follows: B.Z. and H.B.: Conceptualization, Methodology, Writing-reviewing and editing, Visualization, Supervision; H.B.: Project administration, Formal analysis; D.A. and S.A.: Investigation, Data curation, Writing-original draft. All authors have read and approved the published version of the manuscript.
LIST OF ABBREVIATION
| VHN | = Vickers Hardness Number |
| ANOVA | = One-Way Analysis of Variance |
| SD | = Standard Deviation |
| IQR | = Interquartile Range |
| TEGDMA | = Triethylene Glycol Dimethacrylate |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
This study was approved by the Research Ethics Committee of the Faculty of Dentistry at King Abdulaziz University (Jeddah, Saudi Arabia; Approval Number: 141-07-23).
HUMAN AND ANIMAL RIGHTS
All procedures performed in studies involving human participants were in accordance with the ethical standards of institutional and/or research committee and with the 1975 Declaration of Helsinki, as revised in 2013.
CONSENT FOR PUBLICATION
Written informed consent was obtained from all the participants prior to inclusion in the study.
AVAILABILITY OF DATA AND MATERIALS
The data supporting the findings of the article will be available from the corresponding author [H.B] upon reasonable request.
ACKNOWLEDGEMENTS
We would like to thank the Advanced Technology Dental Research Laboratory for providing access to their laboratory and for their valuable contribution to the successful completion of our research project.


